





Medical Diagnostic Products
Lide Laboratories
1081 Hwy 36 E, Suite 6 Maplewood, MN 55109
651-400-7271 www.lidelabs.com info@lidelabs.com
HANSEL is a federal trademark, Tinea Test and Oticell are Minnesota trademarks belonging to Lide Laboratories Inc.
1081 Hwy 36 E, Suite 6 Maplewood, MN 55109
651-400-7271 www.lidelabs.com info@lidelabs.com
HANSEL is a federal trademark, Tinea Test and Oticell are Minnesota trademarks belonging to Lide Laboratories Inc.
POSTIVE RESULTS:
URINE QUAD PLATES
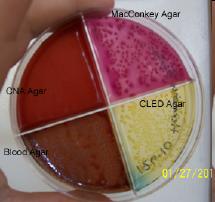
Lide Labs' Urine Quad Plate is designed for the growth, quantification and identification of urinary tract infections. The plate contains four types of media including: CLED Agar (a media designed for the growth and enumeration of organisms in urine), Heart Infusion Blood Agar (for growth of most all organisms and determining hemolysis), CNA Agar (for the promotion of gram-positives at the expense of gram-negatives) and MacConkey Agar (for the growth of gram-negatives with identification). The photo shown to the left is an actual plate used in the determination of a clinical diagnosis. An 8 year old cat was brought to Richland Vet Services with symptoms including blood in the urine and inappropriate urination. As part of the examination, urine was collected and applied to our Urine Quad Plate. The plate was incubated and the following observations were made. The chamber containing Blood Agar showed growth expressed as gray colonies that were hemolytic. CLED Agar showed yellow colonies. MacConkey Agar grew red colonies. And, the CNA Agar showed no growth. Information obtained from the plate was compared against data provided in our Lab ID Chart which contains deferential growth characteristics and morphology of common bacteria on each Urine Quad media. Based on this, the veterinarian was able to determine that this cat had a urine infection due to E.coli bacteria.Special thanks to all at Richland Vet Services (Richland Center, WI) for providing the results and photo to us. NOTE: Use Urine Sediment Stain prior to culturing to identify blood cells, bacteria and other formed elements in urinary sediment at the time of sample collection.
Lide Labs' Urine Quad Plate is designed for the growth, quantification and identification of urinary tract infections. The plate contains four types of media including: CLED Agar (a media designed for the growth and enumeration of organisms in urine), Heart Infusion Blood Agar (for growth of most all organisms and determining hemolysis), CNA Agar (for the promotion of gram-positives at the expense of gram-negatives) and MacConkey Agar (for the growth of gram-negatives with identification). The photo shown to the left is an actual plate used in the determination of a clinical diagnosis. An 8 year old cat was brought to Richland Vet Services with symptoms including blood in the urine and inappropriate urination. As part of the examination, urine was collected and applied to our Urine Quad Plate. The plate was incubated and the following observations were made. The chamber containing Blood Agar showed growth expressed as gray colonies that were hemolytic. CLED Agar showed yellow colonies. MacConkey Agar grew red colonies. And, the CNA Agar showed no growth. Information obtained from the plate was compared against data provided in our Lab ID Chart which contains deferential growth characteristics and morphology of common bacteria on each Urine Quad media. Based on this, the veterinarian was able to determine that this cat had a urine infection due to E.coli bacteria.Special thanks to all at Richland Vet Services (Richland Center, WI) for providing the results and photo to us. NOTE: Use Urine Sediment Stain prior to culturing to identify blood cells, bacteria and other formed elements in urinary sediment at the time of sample collection.

OTICELL™ QUAD PLATES
OTICELL Quad Plates have been designed to help separate and identify the microorganism(s) that are present in an otic sample, especially where a chronic ear problem is evident. This diagnostic information can assist in the selection of medical therapy and allow for additional antimicrobial susceptibility testing where necessary. OTICELL Quad Plates contain four media including: Sabouraud Dextrose Agar with Chloramphenicol (for growth and separate of yeast such as Malasezzia) , Heart Infusion Blood Agar (for growth of most organisms and determining hemolysis), Baird Parker Agar (for growth and separation of staphylococci while inhibiting growth of other organisms) and MacConkey Agar (for the growth of gram-negatives like E coli). A sample was taken from the ear of a two year old goldendoodle that exhibited continual signs of ear irritation. Prior to culturing, the sample was observed microscopically using OTICELL Microbiologic Stain. Yeast cells were identified in the otic sediment. The sample was diluted in Distribution Fluid and applied to each chamber of the OTICELL QUAD Plate. The positive culture results are shown in the photo to the left.
OTICELL Quad Plates have been designed to help separate and identify the microorganism(s) that are present in an otic sample, especially where a chronic ear problem is evident. This diagnostic information can assist in the selection of medical therapy and allow for additional antimicrobial susceptibility testing where necessary. OTICELL Quad Plates contain four media including: Sabouraud Dextrose Agar with Chloramphenicol (for growth and separate of yeast such as Malasezzia) , Heart Infusion Blood Agar (for growth of most organisms and determining hemolysis), Baird Parker Agar (for growth and separation of staphylococci while inhibiting growth of other organisms) and MacConkey Agar (for the growth of gram-negatives like E coli). A sample was taken from the ear of a two year old goldendoodle that exhibited continual signs of ear irritation. Prior to culturing, the sample was observed microscopically using OTICELL Microbiologic Stain. Yeast cells were identified in the otic sediment. The sample was diluted in Distribution Fluid and applied to each chamber of the OTICELL QUAD Plate. The positive culture results are shown in the photo to the left.
TINEA TEST™ Bi-PLATES
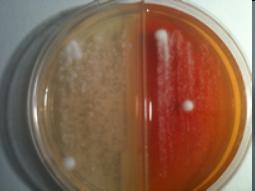
Tinea Test Bi-Plates contain two media types. Chamber I contains Sabouraud Dextrose Agar, a non-selective medium allowing growth of fungi. The agar is included as a positive control. Chamber II contains Dermatophyte Test Medium (DTM), for the selective isolation of dermatophytic fungi. Supplements have been added to the base media to inhibit growth of other organisms. Most dermatophytic fungi will grow on DTM and produce a color change in 3-6 days after inoculation.
The positive plates shown here contain quality control samples of Trichophyton mentagrophytes at 6 days post inoculation. The photo to the left is the top side of the plate showing white growth on the Chamber I and white growth changing to pink on the Chamber II(DTM). The bottom side of the plate shown below, indicates a red color change on DTM. These results are characteristic of dermatophytic fungi on Sabouraund Dextrose and DTM enabling separating and identification. Further idenfication can be made based on morphology using Tinea Test Stain and a microscope.
Tinea Test Bi-Plates contain two media types. Chamber I contains Sabouraud Dextrose Agar, a non-selective medium allowing growth of fungi. The agar is included as a positive control. Chamber II contains Dermatophyte Test Medium (DTM), for the selective isolation of dermatophytic fungi. Supplements have been added to the base media to inhibit growth of other organisms. Most dermatophytic fungi will grow on DTM and produce a color change in 3-6 days after inoculation.
The positive plates shown here contain quality control samples of Trichophyton mentagrophytes at 6 days post inoculation. The photo to the left is the top side of the plate showing white growth on the Chamber I and white growth changing to pink on the Chamber II(DTM). The bottom side of the plate shown below, indicates a red color change on DTM. These results are characteristic of dermatophytic fungi on Sabouraund Dextrose and DTM enabling separating and identification. Further idenfication can be made based on morphology using Tinea Test Stain and a microscope.
